Why do lithium batteries lose charge even when not in use? This natural process, called self-discharge, affects battery life and performance.
Lithium batteries power everything from smartphones to electric vehicles. However, self-discharge can reduce efficiency and shorten lifespan over time.
Understanding self-discharge helps users store, maintain, and use batteries more effectively. It also improves safety and minimizes unnecessary energy loss.
In this post, you’ll learn why lithium battery self-discharge happens, what factors influence it, and how to reduce its impact. Let’s explore practical strategies to extend battery life.

What is Lithium Battery Self-Discharge?
Definition of Self-Discharge
Self-discharge refers to the natural phenomenon where lithium batteries lose their stored energy over time, even when not connected to any device. This internal energy loss occurs while batteries sit unused in storage or remain idle in devices. It represents the battery’s inability to maintain its full charge capacity indefinitely.
Think of it as a slow leak in your battery’s energy tank. Every lithium battery experiences this gradual power drain due to unavoidable internal chemical reactions happening inside its cells.
Understanding the Self-Discharge Process
The self-discharge process in lithium batteries occurs through two primary mechanisms:
Physical Self-Discharge:
- Results from micro short circuits inside battery cells
- Often caused by tiny metal particles or manufacturing defects
- Creates unintended electrical connections between positive and negative electrodes
- This type remains recoverable through recharging
Chemical Self-Discharge:
- Stems from unavoidable chemical reactions between battery components
- Involves electrolyte decomposition and electrode material degradation
- Creates irreversible capacity loss over time
- Accelerates at higher temperatures and states of charge
These internal processes happen continuously regardless of external connections. Both contribute to the battery’s overall self-discharge rate but differ in their recoverability.
Comparing Self-Discharge Rates Across Battery Types
Different battery chemistries exhibit varying self-discharge characteristics. Understanding these differences helps users select appropriate battery technology for specific applications.
| Battery Chemistry | Rechargeable | Self-Discharge Rate |
|---|---|---|
| Lithium-ion | Yes | 2-3% per month |
| Lithium-polymer | Yes | ~5% per month |
| Lead-acid | Yes | 4-6% per month |
| Low self-discharge NiMH | Yes | As low as 0.25% per month |
| Nickel-cadmium | Yes | 15-20% per month |
| Standard NiMH | Yes | 30% per month |
| Lithium metal | No | 10 years shelf life |
| Alkaline | No | 5 years shelf life |
Lithium-ion batteries demonstrate superior self-discharge performance compared to nickel-based alternatives. They lose approximately 2-3% of their charge monthly under optimal storage conditions. This relatively low rate makes them ideal for modern portable electronics and devices requiring reliable standby power.
Temperature significantly impacts these rates. Every 10°C increase roughly doubles the self-discharge speed across all battery types. This explains why batteries stored in hot cars or warm environments lose charge faster than those kept in cool locations.
Key Insight: Lithium-ion batteries offer among the lowest self-discharge rates for rechargeable technologies, outperformed only by specialized low self-discharge NiMH cells specifically designed to address this issue.
Factors Influencing Lithium Battery Self-Discharge Rate
The self-discharge rate of lithium batteries varies significantly based on several key factors. Understanding these elements helps users optimize battery performance and extend their service life.
Temperature
Temperature stands as the most influential factor affecting lithium battery self-discharge rates. It directly impacts internal chemical reaction speeds inside battery cells.
Impact of High and Low Temperatures
- High temperatures (above 30°C/86°F):
- Accelerate chemical reactions within cells
- Double self-discharge rate for every 10°C increase
- Cause permanent capacity loss when prolonged
- May damage battery internal components
- Low temperatures (below 0°C/32°F):
- Slow down self-discharge processes
- Reduce immediate energy loss
- May increase internal resistance
- Could lead to lithium plating when charging
Optimal Storage Temperature Range
Most lithium batteries perform best when stored between 15°C to 25°C (59°F to 77°F). This temperature range minimizes chemical activity while maintaining proper electrolyte conductivity. Relative humidity should remain between 45-75% for ideal conditions.
Pro Tip: Never store lithium batteries in hot cars, direct sunlight, or freezers. These environments dramatically reduce their lifespan through accelerated self-discharge.
Battery Chemistry and Composition
Battery chemistry plays a crucial role in determining self-discharge characteristics. Different lithium technologies exhibit varying stability levels.
Role of Electrode Materials
| Chemistry Type | Electrode Material | Self-Discharge Rate | Notable Properties |
|---|---|---|---|
| LiFePO₄ | Lithium Iron Phosphate | Low (1-2% monthly) | More stable structure, longer cycle life |
| LiCoO₂ | Lithium Cobalt Oxide | Medium (2-3% monthly) | Higher energy density, less stable |
| LiMn₂O₄ | Lithium Manganese Oxide | Medium-High (3-4% monthly) | Better thermal stability than LiCoO₂ |
| NMC | Lithium Nickel Manganese Cobalt | Medium (2-3% monthly) | Balanced performance |
LiFePO₄ batteries demonstrate lower self-discharge rates due to their inherently stable crystal structure. This stability prevents unwanted side reactions between electrodes and electrolyte.
Influence of Electrolyte Stability
Electrolyte composition significantly impacts self-discharge. More stable electrolytes reduce unwanted side reactions in several ways:
- Electrolyte additives improve SEI (Solid Electrolyte Interphase) layer formation
- Higher purity electrolytes contain fewer reactive impurities
- Advanced formulations resist decomposition at higher voltages
- Solid-state electrolytes show promising potential to minimize self-discharge
State of Charge During Storage
The charge level during storage directly influences how quickly lithium batteries self-discharge.
Relationship Between State of Charge and Self-Discharge Rate
Batteries stored at higher charge states (80-100% SOC) experience accelerated self-discharge rates. This occurs because:
- Higher voltages create stronger driving forces for side reactions
- Electrode materials become less stable at extreme charge states
- Electrolyte decomposition increases at elevated potentials
- Internal pressure may rise, stressing battery components
Recommended Storage Charge Level
For optimal long-term storage:
- Best: 40-60% state of charge
- Acceptable: 30-70% state of charge
- Avoid: Below 20% or above 80% state of charge
Maintaining mid-range charge levels during storage minimizes internal stress while providing enough energy to prevent critical discharge. Many manufacturers pre-charge batteries to approximately 50% before shipping for this reason.
Battery Age and Degradation
All lithium batteries age over time, regardless of usage patterns. This calendar aging affects self-discharge behavior.
Effect of Aging on Self-Discharge Rate
As batteries age, they typically experience:
- Gradual increase in self-discharge rates
- Higher internal resistance values
- Reduced capacity retention ability
- Decreased stability of internal components
- More sensitive response to temperature fluctuations
A five-year-old lithium battery might show 2-3× higher self-discharge rates compared to its new condition due to these cumulative aging effects.
Irreversible Capacity Loss
Long-term storage contributes to permanent capacity reduction through:
- SEI layer growth consuming active lithium
- Structural changes in electrode materials
- Electrolyte decomposition products coating active surfaces
- Gradual isolation of active material particles
These changes cannot be reversed through normal charging cycles. Once capacity loss occurs from prolonged storage, it remains permanent.
Manufacturing Quality and Defects
Production quality significantly influences self-discharge performance of lithium batteries.
Impact of Impurities and Defects
Common manufacturing issues increasing self-discharge include:
- Metal particle contamination – Creates micro-short circuits between electrodes
- Separator defects – Allows direct electrode contact points
- Electrode misalignment – Causes uneven current distribution
- Electrolyte impurities – Accelerates unwanted side reactions
- Inadequate formation cycles – Results in unstable SEI layers
Even microscopic metal particles (5-10 μm) can pierce separators or form conductive pathways, dramatically increasing self-discharge rates.
Importance of Quality Control
Premium battery manufacturers implement rigorous quality controls:
- Clean room manufacturing environments (Class 1000-10000)
- Automated optical inspection systems
- X-ray examination for internal defects
- Precise alignment verification
- Extended formation procedures
- Multi-stage testing protocols
- Statistical process control monitoring
These measures identify high self-discharge cells before they reach consumers. Quality screening typically involves voltage monitoring over 7-30 days to detect abnormal discharge patterns.
Measuring Lithium Battery Self-Discharge Rate
Accurate measurement of lithium battery self-discharge remains crucial for quality control, performance evaluation, and reliability assessment. Several methods exist to quantify this phenomenon, each offering different advantages depending on testing requirements.
Open Circuit Voltage (OCV) Method
The OCV method stands as the simplest approach to assess self-discharge rates. It relies on monitoring voltage decay over time when batteries remain disconnected from any load.
Procedure for Measuring Self-Discharge Using OCV
- Initial preparation:
- Fully charge the battery to 100% capacity
- Allow brief stabilization period (1-2 hours)
- Record precise initial voltage (V₀)
- Testing phase:
- Disconnect battery from all loads
- Store under controlled temperature conditions
- Measure voltage at predetermined intervals
- Continue monitoring for specified period (days to weeks)
- Data analysis:
- Plot voltage readings over time
- Calculate voltage drop rate
- Convert to estimated capacity loss
Interpreting Voltage Drop Over Time
Voltage decay doesn’t follow a linear pattern. The relationship between OCV and state of charge follows a battery-specific discharge curve. Interpreting results requires understanding these key points:
- Initial rapid voltage drop (24-48 hours) reflects normal stabilization
- Steady-state decline rate indicates true self-discharge
- Steeper slopes signal faster self-discharge
- Sudden voltage drops suggest potential internal defects
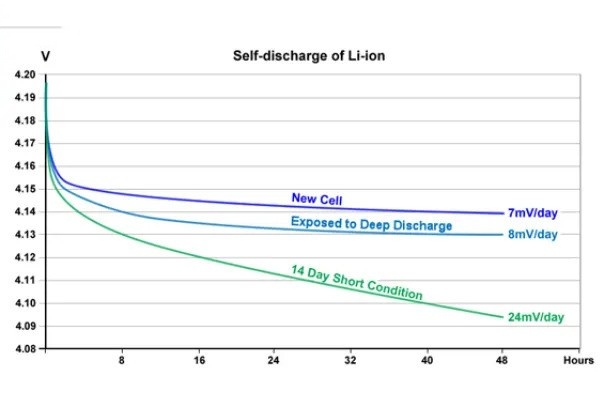
The K-value, representing voltage drop per unit time (mV/day), serves as a standardized metric for comparing self-discharge rates between different batteries.
Example: A high-quality lithium-ion battery might show a K-value of 0.5-1.5 mV/day at room temperature. Values exceeding 3 mV/day could indicate manufacturing defects or degradation issues.
Capacity Test Method
This method directly measures capacity loss during storage periods, providing the most tangible assessment of self-discharge impact on battery performance.

Measuring Self-Discharge Through Capacity Loss
The procedure follows these steps:
- Baseline capacity measurement:
- Fully charge battery to manufacturer specifications
- Discharge at standard rate to cutoff voltage
- Record initial capacity (Q₀) in mAh or Ah
- Storage period:
- Recharge battery completely
- Store under controlled conditions
- Maintain consistent temperature
- Allow predetermined rest time (weeks to months)
- Final capacity measurement:
- Discharge at identical rate to initial test
- Record final capacity (Q₁)
- Document all test parameters
Comparing Initial and Final Capacity Levels
The self-discharge rate calculation uses this formula:
Self-discharge rate = [(Q₀ - Q₁) / Q₀] × 100%
This percentage represents direct capacity loss during the storage period. Manufacturers often specify maximum acceptable self-discharge rates for their products.
| Storage Duration | Acceptable Self-Discharge | Concerning Self-Discharge |
|---|---|---|
| 1 month | 2-3% | >5% |
| 3 months | 5-8% | >12% |
| 6 months | 8-15% | >20% |
| 12 months | 15-25% | >30% |
While highly accurate, this method requires significant testing time. It remains the industry standard for qualification testing despite this drawback.
Coulomb Counting Method
The Coulomb counting approach leverages battery management systems (BMS) to track charge movements into and out of batteries with high precision.
Using BMS to Track Self-Discharge
Modern BMS technology employs specialized measurement circuits to monitor current flow with exceptional accuracy. These systems:
- Measure current at millisecond intervals
- Integrate readings over time (Coulomb counting)
- Account for temperature variations
- Apply compensation algorithms for accuracy
- Store baseline capacity references
This method proves particularly valuable for batteries within electronic devices or energy storage systems equipped with integrated BMS.
Monitoring Coulomb Count During Rest Periods
During rest periods, a properly calibrated BMS can detect the minute current flows indicating self-discharge. The process works through:
- Establishing zero-current baseline when disconnected
- Detecting microscopic current leakage (μA range)
- Accumulating these losses over time
- Converting to capacity loss percentage
The primary advantage comes from real-time monitoring without disconnecting batteries from their systems. This enables ongoing assessment without disrupting normal operation.
High-Precision Measurement Techniques
For research applications, product development, and forensic analysis, several advanced techniques provide deeper insights into self-discharge mechanisms.
Electrochemical Impedance Spectroscopy (EIS)
EIS applies alternating current signals across multiple frequencies to analyze battery internal components and their behaviors.
For self-discharge assessment, EIS offers these insights:
- Identification of specific self-discharge mechanisms
- Detection of micro-short circuits through resistance changes
- Evaluation of electrode interface degradation
- Assessment of SEI layer stability
- Early warning of developing internal issues
The technique requires specialized laboratory equipment but provides unparalleled diagnostic capabilities for understanding the root causes behind self-discharge behaviors.
Potentiostatic Intermittent Titration Technique (PITT)
PITT represents an advanced electrochemical method for precise analysis of battery internal processes. For self-discharge studies, it:
- Applies controlled potentials to battery cells
- Measures resulting current responses over time
- Identifies specific reaction mechanisms
- Quantifies reaction rates for different components
- Distinguishes between physical and chemical self-discharge
This laboratory technique helps battery manufacturers improve designs by pinpointing exact components or materials contributing to self-discharge. The insights gained often lead to formulation adjustments, material substitutions, or manufacturing process improvements.
Industry Application: Leading battery manufacturers use combinations of these techniques in developmental stages. They typically rely on simpler OCV and capacity testing for production quality control due to cost and time constraints.
Impact of Lithium Battery Self-Discharge
Self-discharge affects every lithium battery, influencing performance, safety, and economics. These impacts reach across consumer electronics, electric vehicles, medical devices, and industrial applications.
Reduced Battery Life and Performance
Self-discharge directly diminishes battery performance through several mechanisms, leading to noticeable degradation over time.
Capacity Loss Due to Self-Discharge
Lithium batteries lose capacity through two distinct pathways:
- Temporary capacity loss:
- Occurs from reversible self-discharge
- Recoverable through normal recharging
- Represents energy unavailable until recharged
- Typically accounts for 2-3% monthly loss
- Permanent capacity loss:
- Results from irreversible chemical reactions
- Cannot be recovered through charging
- Accelerates when batteries remain at high or low states of charge
- Compounds with normal cycling degradation
Over time, these effects accumulate. A lithium-ion battery stored for one year might permanently lose 15-20% of its original capacity from self-discharge mechanisms alone.
Shorter Runtime and Decreased Power Output
As self-discharge advances, users experience:
- Reduced runtime between charges
Devices operate for shorter periods before requiring recharging. This effect becomes particularly noticeable in rarely used devices like emergency flashlights or backup power systems. - Diminished peak power delivery
Batteries affected by long-term self-discharge often show increased internal resistance. This resistance limits maximum current output, affecting performance during high-demand situations. - Voltage depression under load
Self-discharged batteries exhibit faster voltage sag when powering devices, potentially triggering early shutdown in voltage-sensitive equipment.
Real-world impact: A smartphone left unused for six months might only provide 4-5 hours of use instead of the expected 8-10 hours, even after full recharging, due to combined effects of self-discharge and permanent capacity loss.
Safety Concerns
Beyond performance issues, self-discharge creates potential safety hazards requiring careful consideration.
Over-Discharge and Cell Damage
When lithium batteries self-discharge below critical voltage thresholds (typically 2.5V-3.0V per cell), several dangerous conditions may develop:
- Copper dissolution
At extremely low voltages, copper current collectors dissolve into the electrolyte, later redepositing as conductive dendrites during recharging. - Structural deformation
Deep discharge causes physical changes in electrode materials, producing mechanical stress and potential separator damage. - SEI layer breakdown
The protective Solid Electrolyte Interphase layer deteriorates during over-discharge, exposing fresh electrode surfaces to unwanted reactions. - Gas generation
Chemical reactions during over-discharge produce gases, causing cell swelling or rupture.
Many of these effects become irreversible once batteries discharge below approximately 2.0V per cell, permanently compromising safety and performance.

Thermal Instability and Fire Hazards
Self-discharge contributes to thermal risks through multiple pathways:
| Risk Factor | Mechanism | Potential Outcome |
|---|---|---|
| Internal short circuits | Dendrite formation from overdischarge | Localized heating, thermal runaway |
| Damaged separators | Physical degradation during deep discharge | Cell shorting, fire, explosion |
| Copper plating | Metal deposition after overdischarge | Increased internal resistance, heating |
| SEI layer breakdown | Loss of protective interface | Exothermic reactions, gas generation |
These conditions become particularly dangerous when over-discharged batteries undergo rapid recharging. The damaged internal structures cannot handle normal charging currents, potentially leading to:
- Excessive heat generation
- Thermal runaway reactions
- Electrolyte vaporization
- Cell rupture or explosion
- Toxic gas release
For these reasons, many battery management systems permanently disable cells detected below critical voltage thresholds.
Maintenance and Replacement Costs
The economic impact of self-discharge extends beyond the battery itself, affecting overall ownership costs.
Frequent Recharging and Battery Replacements
Self-discharge necessitates:
- More frequent charging cycles
Each recharge to compensate for self-discharge counts against the battery’s cycle life, accelerating overall degradation. - Earlier replacement timelines
Batteries experiencing high self-discharge rates typically require replacement 15-30% sooner than expected. - Additional maintenance checks
Systems relying on battery backup require regular verification, increasing labor costs. - Unexpected downtime
Critical equipment may fail when batteries self-discharge below operational thresholds.
These factors compound in large-scale applications. A telecommunications company maintaining thousands of backup battery systems might spend millions annually addressing self-discharge-related issues.
Economic Impact on Battery-Powered Devices
The financial consequences of self-discharge affect different sectors:
Consumer Electronics:
- Reduced resale value of unused devices
- Warranty claims from premature battery failure
- Customer dissatisfaction and brand reputation costs
- Additional service center visits
Electric Vehicles:
- Range anxiety from unpredictable capacity loss
- Decreased resale value from battery degradation
- Higher electricity costs from charging inefficiencies
- Potential early battery replacement (≈$5,000-$20,000)
Industrial Applications:
- Energy storage system inefficiencies
- Backup power reliability concerns
- Increased maintenance requirements
- Operational disruptions from unexpected power loss
These economic impacts make self-discharge a critical consideration in battery selection and system design. Many applications now specify maximum acceptable self-discharge rates in their battery procurement requirements, recognizing long-term financial implications beyond initial purchase costs.
Strategies to Minimize Lithium Battery Self-Discharge
While self-discharge represents an unavoidable aspect of lithium battery chemistry, several practical strategies can significantly reduce its rate and impact. Implementing these approaches extends battery lifespan, improves reliability, and enhances overall performance.
Proper Storage Conditions
Environmental factors dramatically influence self-discharge rates. Creating optimal storage conditions serves as the simplest yet most effective method to preserve battery capacity.
Maintaining Optimal Temperature Range
Temperature directly affects chemical reaction rates inside lithium batteries:
- Ideal storage temperature: 15°C to 25°C (59°F to 77°F)
- Acceptable range: 10°C to 30°C (50°F to 86°F)
- Avoid: Temperatures above 35°C (95°F) or below 0°C (32°F)
Every 10°C increase approximately doubles self-discharge rate. Consumers should store batteries in climate-controlled environments whenever possible. Basements often provide naturally stable temperatures suitable for battery storage.
Pro Tip: When storing multiple batteries, avoid stacking them together. This prevents heat buildup between units and allows better air circulation.
Avoiding Extreme Temperatures and Humidity
Beyond temperature, other environmental factors require attention:
- Humidity control: Maintain relative humidity between 45-75%
- Direct sunlight: Shield batteries from UV exposure
- Heat sources: Keep batteries away from heaters, furnaces, or hot appliances
- Freezing conditions: Never store lithium batteries in freezers or unheated garages during winter
High humidity can penetrate battery casings over time, promoting internal corrosion and increasing self-discharge pathways. Conversely, extremely dry conditions might accelerate electrolyte evaporation in some battery designs.
Partial Charge Storage
The battery’s state of charge during storage significantly impacts self-discharge rates and long-term health.
Storing Batteries at 40-60% State of Charge
Research demonstrates clear advantages to partial-charge storage:
| State of Charge | Self-Discharge Rate | Capacity Loss After 1 Year | Recommendation |
|---|---|---|---|
| 100% (Full) | High | 15-25% | Avoid for storage |
| 70-80% | Medium | 10-15% | Acceptable for short-term |
| 40-60% | Low | 5-10% | Optimal for long-term |
| 20-30% | Very Low | 10-15% | Risk of over-discharge |
| 0-10% | Critical | 20-30%+ | Severe damage risk |
The 40-60% charge range represents the ideal balance point. This level provides:
- Minimized chemical stress on electrode materials
- Reduced electrolyte degradation
- Lower internal pressure within cells
- Sufficient reserve to prevent critical discharge
Balancing Self-Discharge and Capacity Retention
Implementing optimal charge levels requires understanding the trade-offs:
- Immediate use priority: Charge to 80-90% for devices needed soon
- Medium-term storage (1-3 months): Charge to 50-60%
- Long-term storage (4+ months): Maintain at 40-50%
- Unknown duration: Aim for 40% with periodic verification
For practical implementation, most devices display approximate charge percentages. When preparing batteries for storage:
- Charge or discharge to target level
- Allow 2-3 hours for voltage stabilization
- Verify charge level again before storage
- Recheck every 3-6 months during long-term storage

Battery Management Systems (BMS)
Modern lithium battery applications increasingly rely on intelligent management systems to mitigate self-discharge impacts.
Role of BMS in Monitoring and Controlling Self-Discharge
A battery management system serves as the guardian against excessive self-discharge through several functions:
- Continuous monitoring: Tracking voltage, temperature, and current
- Early detection: Identifying abnormal self-discharge patterns
- Automatic intervention: Initiating corrective measures when needed
- Data logging: Recording battery health metrics for analysis
- User alerts: Providing timely warnings about potential issues
Advanced BMS designs can detect self-discharge rates exceeding normal parameters, often signaling internal defects requiring attention. This early warning capability prevents cascading failures affecting entire battery packs.
Cell Balancing and Over-Discharge Protection
In multi-cell battery packs, individual cells naturally develop slight variations in self-discharge rates. These differences compound over time, creating potentially dangerous imbalances.
The BMS addresses this through:
Passive Cell Balancing:
- Bleeding excess energy from higher-charged cells
- Equalizing voltage across the pack
- Preventing overcharge of individual cells
Active Cell Balancing:
- Redistributing energy between cells
- Maximizing available capacity
- Reducing wasted energy
Over-Discharge Prevention:
- Monitoring individual cell voltages
- Disconnecting loads before critical discharge
- Entering sleep modes to preserve essential functions
- Preventing irreversible damage from complete depletion
These protective functions prove especially valuable in large battery systems like those found in electric vehicles or home energy storage units, where cell count increases complexity.
Advances in Battery Chemistry and Manufacturing
Research continues to yield improvements specifically targeting self-discharge reduction.
Development of Low Self-Discharge Materials
Material science advances have produced several promising developments:
- Modified cathode coatings:
- Aluminum oxide surface treatments
- Nano-scale protective layers
- Reduced reactivity with electrolytes
- Enhanced separator materials:
- Ceramic-reinforced polymers
- Multi-layer designs
- Improved ion selectivity
- Stabilized electrolyte formulations:
- Novel additives preventing decomposition
- Reduced flammability
- Extended electrochemical stability windows
- Next-generation anodes:
- Silicon-carbon composites
- Lithium titanate options
- Reduced solid electrolyte interphase formation
These advancements progressively reduce self-discharge rates while simultaneously improving safety profiles and overall battery performance.
Improved Manufacturing Processes and Quality Control
Manufacturing refinements play equally important roles in minimizing self-discharge:
- Cleanroom production environments Ultra-clean facilities eliminate contaminants causing micro-short circuits
- Precision electrode alignment Advanced machinery ensures perfect positioning of battery components
- Laser inspection systems Automated detection of microscopic defects invisible to human operators
- Extended formation protocols Longer conditioning cycles establish more stable internal structures
- Multi-stage testing regimens Comprehensive validation identifies potential self-discharge issues
- Statistical process control Real-time monitoring ensures consistent quality across production batches
Quality manufacturers now implement “K-value testing” during production. This procedure measures voltage decay rates over shorter periods, allowing identification of batteries predisposed to high self-discharge before shipping to customers.
Industry Trend: Leading manufacturers increasingly publish self-discharge specifications in technical documentation, recognizing its importance for applications requiring long-term reliability.
Best Practices for Lithium Battery Users
Proper care extends battery life, improves performance, and enhances safety. Following best practices minimizes self-discharge, reduces wear, and prevents hazards.
Regular Battery Maintenance
Periodic Charging and Discharging Cycles
Regular charging prevents deep discharge. Letting a lithium battery sit at low voltage shortens lifespan. A controlled charge-discharge cycle maintains efficiency.
Monitoring Battery Health and Performance
Track battery voltage and capacity. Sudden drops signal degradation. Use battery management systems (BMS) or built-in monitoring tools for real-time performance checks.
Proper Charging Techniques
Using Compatible Chargers and Following Manufacturer Guidelines
Each battery type requires a specific charger. Mismatched chargers cause inefficiencies or overheating. Manufacturer recommendations ensure optimal voltage and current.
Avoiding Overcharging and Deep Discharge
Overcharging stresses battery chemistry. Deep discharge leads to irreversible capacity loss. Keep charge levels between 20% and 80% for longevity.
Safe Handling and Disposal
Protecting Batteries from Physical Damage
Crushed or punctured batteries risk short circuits. Store them in protective cases. Avoid exposure to extreme heat or water.
Disposing of Batteries Responsibly and Recycling When Possible
Old batteries contain hazardous materials. Recycling prevents pollution. Many retailers and recycling centers accept used lithium batteries.
| Best Practice | Benefit |
|---|---|
| Periodic Charging | Extends lifespan, prevents deep discharge |
| Voltage Monitoring | Detects performance decline early |
| Proper Chargers | Ensures efficiency, prevents overheating |
| Controlled Charging | Reduces stress, prevents degradation |
| Safe Storage | Prevents physical damage, short circuits |
| Recycling | Protects environment, complies with laws |
Adopting these habits keeps lithium batteries efficient and safe. A proactive approach ensures long-term reliability while reducing risks.
Conclusion
Lithium battery self-discharge reduces capacity, weakens performance, and shortens lifespan. Understanding its causes helps users minimize energy loss.
Proper charging, regular maintenance, and safe handling extend battery life. Monitoring voltage prevents over-discharge, protecting cells from damage.
Managing self-discharge ensures longer runtime, better efficiency, and improved safety. Following best practices reduces costs and environmental impact.
By applying these strategies, users can maximize lithium battery performance while enhancing reliability and safety.
FAQs
Q: What is the typical self-discharge rate of lithium-ion batteries?
A: Lithium-ion batteries typically experience a self-discharge rate of 2-3% per month under normal conditions. This rate positions them favorably compared to other rechargeable technologies such as nickel-cadmium (15-20% per month) or standard NiMH (30% per month). Environmental factors, particularly temperature, can significantly affect this rate.
Q: How can I tell if my lithium battery has experienced significant self-discharge?
A: Signs of significant self-discharge include unexpectedly short runtime, devices shutting down prematurely, difficulty holding charge, and requiring more frequent recharging. Physical indicators might include battery warming during charging, swelling, or voltage readings below normal levels. Modern devices often display battery health metrics showing capacity loss. Using a voltmeter to check resting voltage can also indicate excessive self-discharge.
Q: Is it safe to use a lithium battery that has been stored for an extended period?
A: Safety depends on storage conditions and remaining voltage. If a lithium battery has self-discharged below 2.5V per cell, it may develop internal damage. Check for physical swelling, unusual heat, or odors before use. Measure voltage if possible. Batteries maintaining above 3.0V per cell are generally safe. Those below critical thresholds should be recycled rather than recharged due to potential fire risks.
Q: Can self-discharge be completely eliminated in lithium batteries?
A: Self-discharge cannot be completely eliminated in lithium batteries. The article specifically states this reaction is “unavoidable” due to fundamental electrochemical processes occurring inside the cells. Even with perfect manufacturing and optimal storage conditions, some self-discharge will occur. However, manufacturers continue developing technologies to minimize its rate through improved materials, better production processes, and advanced battery management systems.
Q: How often should I recharge my lithium batteries to minimize self-discharge?
A: For long-term storage, check and recharge lithium batteries every 3-6 months to prevent deep discharge. Maintain charge levels between 40-60% rather than full capacity. For infrequently used devices, a quarterly recharging schedule maintains battery health. In critical applications like medical equipment or emergency systems, monthly verification ensures reliability. Never let stored lithium batteries discharge below critical voltage thresholds.
