Imagine your phone suddenly heating up, smoking, and catching fire. This is the danger of thermal runaway in lithium-ion batteries.
Thermal runaway happens when a battery generates more heat than it can release. This chain reaction can lead to extreme temperatures, fires, or explosions. Understanding this risk is crucial for safety, especially as lithium-ion batteries power everything from smartphones to electric vehicles.
In this post, you’ll learn what causes thermal runaway, why it happens, and how to prevent it. Whether you’re an engineer or a consumer, this knowledge helps protect both devices and lives.

What is Thermal Runaway?
Thermal runaway is a dangerous phenomenon in lithium-ion batteries where an uncontrollable, self-sustaining increase in temperature occurs. This critical failure can transform a reliable power source into a serious safety hazard, potentially resulting in battery fires or explosions.
Explanation of the Chain Reaction
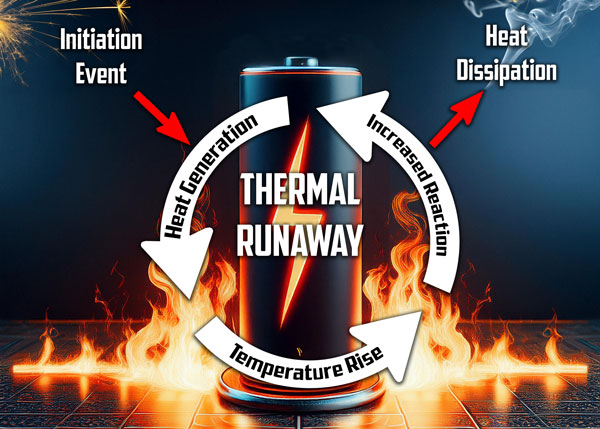
Thermal runaway operates on a positive feedback loop principle—where each step in the process accelerates the next, creating a rapid, cascading effect that’s extremely difficult to stop once initiated.
The process follows this dangerous sequence:
- Initial heating – Heat generation begins from internal or external sources
- Accelerated chemical reactions – Rising temperatures speed up reactions within the battery
- Increased heat production – Faster reactions generate more heat
- Venting of gases – Pressure builds, causing the release of flammable and toxic gases
- Further temperature increase – Heat dissipation can’t keep pace with heat generation
- Catastrophic failure – Temperature skyrockets to 600°C+ within minutes, potentially resulting in fire or explosion
What makes thermal runaway particularly dangerous is its self-sustaining nature. Even after removing the initial heat source, the battery continues to generate heat and toxic gases, creating oxygen and fueling further reactions.
Conditions Leading to Thermal Runaway
Several factors can trigger the thermal runaway process in lithium-ion batteries:
| Trigger | Description | Relative Risk |
|---|---|---|
| Overcharging | Pushing a battery beyond its safe maximum voltage | High |
| Multiple overdischarges | Repeatedly draining below recommended thresholds | Medium |
| Rapid charging | Excessive current flow during fast charging | Medium-High |
| Short circuits | Internal or external electrical shorts | Very High |
| Extreme temperatures | Operation outside safe temperature range | High |
| Physical damage | Crushing, puncturing, or other mechanical trauma | High |
| Manufacturing defects | Quality issues during production | Variable |
The probability of thermal runaway increases with battery size and number of cells, making larger battery systems like those in electric vehicles statistically more susceptible to failure compared to smaller devices like smartphones.
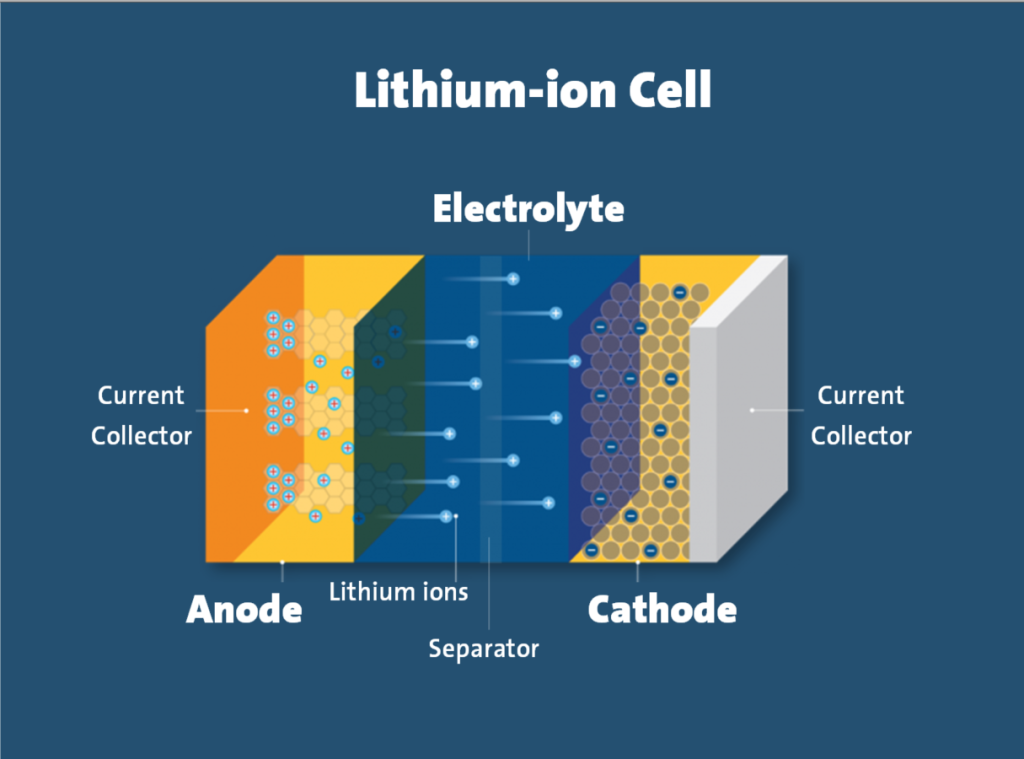
Why Lithium-Ion Batteries Are Susceptible
Lithium-ion batteries possess inherent characteristics that make them particularly vulnerable to thermal runaway:
- High energy density – Their compact, energy-rich design means more stored energy that can be released during failure
- Reactive components – Lithium is highly reactive with strong reducing potential
- Flammable electrolytes – The alkyl carbonate electrolytes used are combustible
- Self-oxidizing – Cathode materials can release oxygen during degradation, fueling internal combustion
- Thin separators – The physical barrier between positive and negative electrodes is delicate and can be compromised
When thermal runaway occurs in lithium-ion batteries, temperatures can reach up to 1100°C (2012°F), releasing dozens of toxic gases including hydrogen fluoride (HF), carbon monoxide (CO), and various volatile organic compounds.
Perhaps most concerning is that lithium-ion battery fires are self-sufficient and continue to burn without external oxygen. This makes traditional firefighting approaches that rely on oxygen deprivation less effective, presenting unique challenges for emergency responders.
Progression of Thermal Runaway
Understanding how thermal runaway progresses in lithium-ion batteries is crucial for developing effective safety measures and prevention strategies. This dangerous phenomenon follows a predictable sequence of events that can rapidly escalate from normal operation to catastrophic failure.
Heat Generation in Normal Operation
During regular operation, lithium-ion batteries naturally generate small amounts of heat as a byproduct of the electrochemical processes converting chemical energy into electrical energy. This normal heat generation is typically manageable and expected:
- Small temperature increases (5-10°C above ambient) during charging
- Slightly elevated temperatures during high-drain applications
- Brief warming during rapid charge/discharge cycles
Most users have experienced this normal warming when using laptops, smartphones, or other battery-powered devices for extended periods. Modern devices incorporate thermal management systems designed to handle this expected heat generation.
Abuse Scenarios Leading to Excessive Heat
When batteries experience conditions beyond their design parameters, heat generation can increase dramatically. Common abuse scenarios include:
| Abuse Type | Examples | Heat Impact |
|---|---|---|
| Thermal | Exposure to high ambient temperature (>35°C) | Accelerates internal reactions |
| Electrical | Overcharging, rapid charging, short circuits | Direct conversion of electrical energy to heat |
| Mechanical | Crushing, puncturing, internal separator damage | Creates internal short circuits |
| Manufacturing | Contaminants, improper assembly, poor quality control | Reduces thermal stability margins |
The balance between heat generation and heat dissipation determines whether a battery remains in safe operating conditions or enters the dangerous territory of thermal runaway.
Positive Feedback Loop of Heat Generation
When heat generation exceeds dissipation, a positive feedback loop begins:
- Temperature rises beyond normal levels.
- Electrolyte decomposition releases flammable gases.
- Separator failure causes internal short circuits.
- Oxygen release from the cathode accelerates combustion.
- More heat is generated, fueling a self-sustaining cycle.
This runaway reaction rapidly escalates within seconds to minutes, making intervention difficult.
Temperature Escalation and Self-Sustaining Cycle
Once the positive feedback loop initiates, temperature escalation can become rapid and self-sustaining:
- SEI layer breakdown – The protective Solid Electrolyte Interface layer decomposes
- Electrolyte breakdown – Flammable organic solvents begin to decompose and vaporize
- Separator failure – The physical barrier between electrodes melts or ruptures
- Anode/cathode reactions – Direct reactions between battery components release significant heat
- Cathode decomposition – Metal oxide cathodes release oxygen, fueling combustion
- Gas generation and venting – Pressure buildup forces the release of flammable and toxic gases
This cycle can escalate from concern to catastrophe in minutes as each stage triggers and accelerates the next.

Key Temperature Thresholds
Different temperature ranges represent critical transition points in the thermal runaway process:
Normal Safe Operating Range
- 20°C to 45°C (68°F to 113°F): Ideal operating temperature range
- 0°C to 60°C (32°F to 140°F): Extended operating range with reduced performance
Onset of Instability (Above 60°C)
- 60°C to 80°C (140°F to 176°F): Concern threshold, accelerated aging
- 80°C to 120°C (176°F to 248°F): SEI layer begins decomposition
- 120°C to 200°C (248°F to 392°F): Separator begins melting, electrolyte breakdown initiates
Thermal Runaway Ignition
- 200°C to 250°C (392°F to 482°F): Exothermic reactions accelerate dramatically
- Above 500°C (932°F): Full thermal runaway with potential ignition
- Up to 1100°C (2012°F): Peak temperatures during thermal runaway events
Timeframe for Battery Fire or Explosion
The progression from initial heat buildup to catastrophic failure can vary dramatically depending on battery size, design, and the specific trigger:
- Early warning signs: Minutes to hours (venting gases, swelling, unusual heat)
- From thermal runaway initiation to fire: Seconds to minutes
- From first visible smoke to full involvement: Often less than 30 seconds
What makes lithium-ion battery fires particularly dangerous is their self-sustaining nature. Even after initial extinguishment, the internal chemical reactions and heat generation can continue, leading to potential reignition hours after the initial event.
The rapid progression of thermal runaway emphasizes why early detection systems and proper battery management are critical for preventing these potentially devastating events.
Gases Released During Thermal Runaway
When a lithium-ion battery undergoes thermal runaway, it doesn’t just generate extreme heat—it also releases a complex mixture of gases that pose significant health and safety risks.
Types of Released Gases
The chemical reactions during thermal runaway produce dozens of different gaseous compounds, varying in both composition and concentration depending on the battery chemistry, state of charge, and stage of thermal runaway. These gases can be categorized into several major groups:
Toxic Gases (HF, HCl, HCN, SO₂, NH₃)
Perhaps the most concerning emissions are the highly toxic gases that can cause immediate health effects even in small concentrations:
| Gas | Source | Health Effects | Concentration Range |
|---|---|---|---|
| Hydrogen Fluoride (HF) | Decomposition of LiPF₆ electrolyte salt | Severe respiratory damage, skin burns | Up to hundreds of ppm |
| Hydrogen Chloride (HCl) | Breakdown of chlorinated components | Respiratory irritation, pulmonary edema | Variable |
| Hydrogen Cyanide (HCN) | Thermal decomposition of nitrogen-containing materials | Rapid asphyxiation, cellular hypoxia | Low ppm range |
| Sulfur Dioxide (SO₂) | Degradation of sulfur-containing components | Respiratory irritation, bronchoconstriction | Variable |
| Ammonia (NH₃) | Decomposition of nitrogen compounds | Eye and respiratory irritation | Variable |
HF is particularly hazardous as concentrations can reach hundreds of parts per million (ppm) near a battery in thermal runaway—far exceeding occupational exposure limits and potentially causing severe respiratory damage.
Flammable Gases (CO, C₂H₄, CH₄, H₂)
The thermal decomposition process releases several highly flammable gases that contribute to fire risk:
- Carbon Monoxide (CO): Can reach percentage-level concentrations (tens of thousands of ppm)
- Ethylene (C₂H₄): Highly flammable hydrocarbon
- Methane (CH₄): Common combustible gas
- Hydrogen (H₂): Extremely flammable with wide explosive range
These gases not only present inhalation hazards but significantly increase the risk of fire and explosion, creating a secondary hazard beyond the initial thermal event.
Carcinogenic Compounds (Formaldehyde)
Several carcinogenic volatile organic compounds (VOCs) may be released during thermal runaway:
- Formaldehyde: A known human carcinogen
- Various aromatic hydrocarbons
- Partially oxidized organic compounds
Long-term exposure to these compounds poses chronic health risks beyond the acute dangers of the thermal event itself.
Combustion Products (CO₂)
As thermal runaway progresses to fire, standard combustion products appear:
Primary combustion products:
├── Carbon dioxide (CO₂) - Major product
├── Water vapor (H₂O) - From hydrogen-containing compounds
└── Particulate matter - Smoke and soot containing various compounds
CO₂ levels typically rise significantly only after flammable gases have ignited, marking the transition from thermal decomposition to active combustion.
Gas Release Patterns at Different Stages
The emission profile changes dramatically as thermal runaway progresses:
- Early warning stage (80-120°C):
- Initial release of HF and CO can occur
- These gases may serve as early indicators of impending thermal runaway
- Concentration levels begin to rise above background
- Pre-runaway stage (120-200°C):
- Electrolyte breakdown begins
- Release of carbonate solvents (DMC, EMC, EC)
- Venting of early decomposition products
- Active runaway stage (>200°C):
- Rapid release of flammable gases
- Significant production of toxic gases
- Potential ignition of vented gases
- Combustion stage (500-1100°C):
- Dominated by traditional combustion products
- High levels of particulate matter
- Continued generation of toxic gases
Notably, monitoring gases like HF and CO can potentially provide early warning of thermal events, as their concentrations may rise notably before visible signs of thermal runaway appear.
Health and Environmental Risks
The gaseous emissions from thermal runaway present multifaceted risks:
Immediate Health Hazards
- Acute toxicity: Gases like HF and HCN can cause severe respiratory damage or death at relatively low concentrations
- Asphyxiation risk: Displacement of oxygen by CO₂ and CO
- Burns and trauma: From ignition of flammable gases
- Visibility impairment: Smoke and particulates can hinder evacuation
Broader Impact Zone
The danger zone from toxic gases extends significantly beyond the immediate fire area. While fire and explosion risks are generally confined to the vicinity of the battery, toxic gases can:
- Spread through ventilation systems
- Permeate throughout enclosed spaces
- Reach potentially fatal concentrations in poorly ventilated areas
- Persist after the thermal event is controlled
Environmental Concerns
- Air quality degradation: Persistent plumes of toxic gases
- Water contamination: Runoff from firefighting efforts can carry dissolved toxic compounds
- Soil contamination: Deposition of hazardous materials
The complex mixture of gases produced during thermal runaway necessitates comprehensive safety systems, proper ventilation design, and specialized emergency response protocols for facilities managing large-scale battery systems.
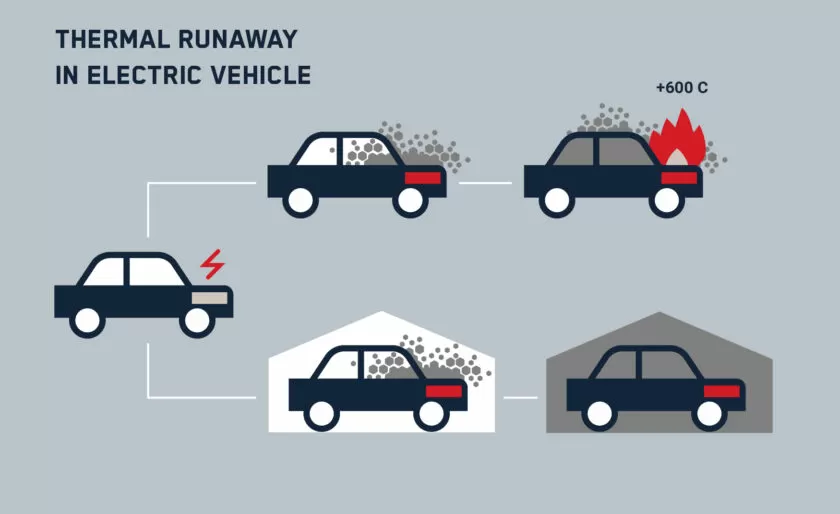
Real-Life Risks and Consequences of Thermal Runaway
Thermal runaway in lithium-ion batteries is not just a theoretical issue—it poses serious real-world risks. As battery technology advances and energy storage systems grow larger, the consequences of thermal runaway become more severe.
Increased Risk with Larger Battery Packs (EVs, Energy Storage)
Larger battery systems, such as those in electric vehicles (EVs), energy storage facilities, and industrial applications, present higher risks due to:
- More Cells, More Failure Points – A single failing cell can trigger a chain reaction in an entire battery pack.
- Higher Energy Density – Large-scale batteries store massive amounts of energy, making thermal runaway more severe.
- Limited Heat Dissipation – Dense battery configurations make cooling difficult, increasing the likelihood of overheating.
Example:
EV battery fires, like those seen in Tesla and Chevy Volt incidents, highlight the challenge of controlling multi-cell battery failures.
Fire and Explosion Hazards
Lithium-ion battery fires are fast-spreading and intense due to high temperatures and flammable materials inside the battery.
| Risk Factor | Impact on Fire Behavior |
|---|---|
| High Heat Release | Fires reach 500-1000°C, igniting nearby materials. |
| Oxygen Generation | The battery self-sustains combustion without external oxygen. |
| Flammable Electrolyte | Organic solvents fuel violent flames and explosions. |
Once a lithium battery ignites, it can be nearly impossible to stop without specialized suppression techniques.
Difficulty in Extinguishing Battery Fires
Traditional fire extinguishing methods fail against lithium-ion battery fires due to:
- Self-Sustaining Reactions – Battery cells continue to generate heat even after flames appear extinguished.
- Re-Ignition Risks – Even if put out, a battery fire can restart hours or days later.
- Limited Effectiveness of Water – Water can cool the battery but does not stop thermal runaway.
Best Practices for Fire Suppression: ✅ Use specialized extinguishers (e.g., Class D, NOVEC gas, or FST aerosol).
✅ Flood EV battery fires with water for extended cooling.
✅ Monitor for re-ignition after extinguishing.
Toxic Gas Inhalation Risks
Thermal runaway releases dozens of hazardous gases, many of which are highly toxic.
Common Gases Released During Thermal Runaway:
| Gas | Health Impact |
|---|---|
| HF (Hydrogen Fluoride) | Can cause severe respiratory damage and skin burns. |
| CO (Carbon Monoxide) | Highly poisonous—can lead to suffocation. |
| HCl (Hydrogen Chloride) | Causes eye and lung irritation. |
| Formaldehyde | Carcinogenic, long-term exposure can cause cancer. |
Even small battery fires can create dangerous air conditions, requiring protective gear for responders.
Long-Term Impact on First Responders and Environment
Battery fires and explosions have lasting effects on both emergency personnel and the environment.
- Risk to First Responders
- Toxic fume exposure can cause permanent lung damage.
- Fires in enclosed spaces (e.g., parking garages) pose higher danger levels.
- Lack of standardized training for handling EV battery fires increases risks.
- Environmental Consequences
- Toxic gas emissions contribute to air pollution.
- Battery fire residues contaminate soil and water supplies.
- Recycling challenges for burned lithium-ion batteries remain unresolved.
Methods to Prevent Thermal Runaway
As lithium-ion batteries continue to power more aspects of our lives, from smartphones to electric vehicles and grid storage systems, preventing thermal runaway has become a critical safety concern. Fortunately, researchers, manufacturers, and regulatory bodies have developed multiple approaches to mitigate these risks.
Battery Design and Material Improvements
Modern battery design focuses on incorporating inherently safer materials and components to reduce the likelihood of thermal events.
Advanced Cathode Materials (LiFePO₄ vs. LiCoO₂)
The choice of cathode material significantly impacts battery safety profiles:
| Cathode Material | Energy Density | Safety Profile | Thermal Stability | Applications |
|---|---|---|---|---|
| LiCoO₂ (LCO) | High | Moderate to Poor | Oxygen release >200°C | Consumer electronics |
| LiFePO₄ (LFP) | Moderate | Excellent | Minimal oxygen release | EVs, grid storage |
| LiMn₂O₄ (LMO) | Moderate | Good | Better than LCO | Medical devices, power tools |
| NMC* | High | Fair | Varies by formulation | EVs, portable devices |
*NMC = Lithium Nickel Manganese Cobalt Oxide
LiFePO₄ cathodes significantly reduce thermal runaway risks due to their stable olivine structure that prevents oxygen release even at elevated temperatures, making them increasingly popular for applications where safety is paramount.
Self-Sealing Separators
Advanced separator technologies provide crucial protection against internal short circuits:
- Shutdown separators: Designed to melt and close pores at elevated temperatures (typically 130°C), halting ion flow before thermal runaway
- Ceramic-coated separators: Provide mechanical strength and heat resistance
- Multi-layer designs: Combine different materials for enhanced protection
These technologies create a fail-safe that can prevent a localized issue from cascading into full thermal runaway.
Flame Retardant Electrolytes
Reducing electrolyte flammability addresses one of the primary hazards during thermal events:
Flame retardant approaches:
├── Phosphorus-containing additives
│ └── Inhibit combustion through radical scavenging
├── Fluorinated components
│ └── Reduce vapor pressure and flammability
└── Non-flammable ionic liquid electrolytes
└── Higher thermal stability threshold
While these modifications often come with performance trade-offs, they provide critical safety enhancements for applications where risk mitigation is paramount.
Battery Management System (BMS)
The BMS serves as the brain of the battery system, providing intelligent monitoring and control functions.

Monitoring Cell Voltage, Current, and Temperature
Modern BMS systems continuously monitor critical parameters:
- Voltage monitoring: Cell-level measurement to detect anomalies (±5mV precision)
- Current monitoring: Real-time tracking of charge/discharge rates
- Temperature sensing: Strategic placement of sensors to detect hotspots
- Impedance measurement: Tracking internal resistance changes as potential failure indicators
For lithium-ion systems, temperature monitoring is particularly critical, as it provides the earliest indication of potential thermal issues.
Overcharge and Overdischarge Protection
The BMS implements voltage limits to prevent damage:
- Upper voltage limit enforcement: Prevents cathode degradation and lithium plating
- Lower voltage threshold protection: Prevents anode copper dissolution and structural damage
- Current limitation: Restricts charge/discharge rates based on temperature and state of charge
- Emergency disconnection: Physically breaks the circuit if critical limits are exceeded
These protections address the most common electrical triggers of thermal runaway.
Balancing and Fault Detection
Advanced BMS functions ensure the battery system operates within safe parameters:
- Cell balancing to prevent individual cells from exceeding voltage limits
- Isolation fault detection to identify insulation breakdowns
- Short circuit protection with fast-acting fuses or circuit breakers
- Ground fault monitoring to detect unintended electrical paths
- Early warning algorithms that recognize patterns preceding thermal events
In large battery packs, sophisticated BMS systems can identify and isolate problematic cells before they impact the entire system.
Storage and Handling Guidelines
Proper storage and handling practices significantly reduce thermal runaway risks throughout a battery’s lifecycle.
Proper Temperature Control (Ideal Range: 1.7°C – 32°C)
Temperature management is critical for battery safety:
- Ideal storage temperature: 15°C to 25°C (59°F to 77°F)
- Acceptable range: 1.7°C to 32°C (35°F to 90°F)
- Avoid extreme cold: Below 0°C can lead to lithium plating during charging
- Avoid high heat: Above 32°C accelerates aging and increases thermal instability
Climate-controlled environments for battery storage provide significant safety benefits and extend battery life.
Avoiding Physical Damage
Physical protection prevents mechanical triggers for thermal runaway:
- Robust packaging to prevent puncture, crushing, or impact
- Proper mounting to minimize vibration and stress
- Protection from water and humidity exposure
- Avoidance of conductive tools or materials that could cause short circuits
- Regular inspection for signs of swelling, leaking, or damage
Educating users about proper handling is particularly important for consumer-level applications.
Safe Battery Disposal and Recycling
End-of-life management reduces risks from degraded batteries:
- Replace lithium-ion batteries that reach 80% of original capacity
- Use specialized battery recycling services rather than general waste disposal
- Discharge batteries to approximately 30% before disposal
- Isolate terminals with non-conductive tape to prevent short circuits
- Follow local regulations for hazardous material handling
Proper disposal not only prevents potential thermal events but also enables valuable material recovery.
Thermal Barriers and Insulation
Physical barriers prevent thermal propagation between cells and contain thermal events when they occur.
Go-Therm Thermal Runaway Barriers
Specialized materials designed for battery applications provide critical protection:
- Go-Therm 150: Glass one-sided silicone laminate for interior battery pack lining
- Go-Therm 315: Fiberglass dual-backed insulation for module-to-module barriers
- Key functions include:
- Containment of ejecta during venting
- Prevention of flame propagation between cells
- Thermal isolation to prevent cascading failures
- Reduction of heat transfer to adjacent components
These materials are specifically engineered for the extreme conditions of battery thermal events.
Pyrel-Therm Insulation Materials
Advanced heat shield materials offer specialized properties:
- Pyrel-Therm EIG 1000: Excellent thermal resistance with minimal shrinkage
- Pyrel-Therm ES 1100: Compression-resistant with low thermal conductivity
- Pyrel-Therm RMC Mica Heat Shield: Very low heat transfer and effective gas barrier
- Pyrel-Therm TS 800C: Superior resistance to flames and high temperatures
These specialized materials can maintain integrity at temperatures exceeding 800°C, providing critical containment during thermal events.
Regulatory Compliance and Industry Standards
Adherence to established standards ensures consistent safety across the industry.
UL 9540A and UL 1973 Certifications
Key safety certifications provide standardized testing frameworks:
- UL 1973: Standard for Batteries for Use in Stationary, Vehicle Auxiliary Power and Light Electric Rail Applications
- Tests cell and battery electrical safety
- Evaluates thermal cycling endurance
- Verifies mechanical shock resistance
- Confirms proper thermal management
- UL 9540A: Test Method for Evaluating Thermal Runaway Fire Propagation in Battery Energy Storage Systems
- Assesses thermal runaway propagation behavior
- Evaluates gas composition during thermal events
- Measures heat release and fire-fighting challenges
- Determines effectiveness of protection measures
These certifications are increasingly becoming mandatory for large-scale battery installations.
IEEE Battery Safety Recommendations
Industry best practices provide additional guidance:
- IEEE 1625: Standard for Rechargeable Batteries for Mobile Computing
- IEEE 1725: Standard for Rechargeable Batteries for Cellular Telephones
- IEEE 450: Recommended Practice for Maintenance, Testing, and Replacement of Vented Lead-Acid Batteries
- Key recommendations include:
- Bi-annual preventative maintenance visits for VRLA batteries
- Regular capacity testing to identify degradation
- Comprehensive monitoring system installation
- Documentation of battery performance history
Following these standards significantly reduces thermal runaway risks across all applications of lithium-ion technology.
Fire Suppression and Extinguishing Methods
Lithium-ion battery fires are exceptionally difficult to control due to their self-sustaining nature. Unlike conventional fires, they continue generating heat even after the flames appear extinguished. This section explores the challenges of lithium-ion battery fires, why traditional firefighting methods fail, and effective suppression techniques.
Challenges of Lithium-Ion Battery Fires
Unlike typical fires, lithium-ion battery fires have unique hazards that make them extremely difficult to extinguish:
- Self-Sustaining Reactions – Batteries continue generating heat due to exothermic chemical reactions.
- Re-Ignition Risk – Even after being extinguished, battery cells can spontaneously reignite hours or days later.
- Toxic Gas Emissions – Fires release hazardous gases like hydrogen fluoride (HF), carbon monoxide (CO), and other carcinogens.
- Explosive Potential – Overheated batteries can rupture violently, spreading flames and debris.
Why Traditional Firefighting Methods Are Ineffective
Conventional firefighting approaches do not work effectively against lithium-ion battery fires:
| Method | Why It Fails |
|---|---|
| Water | Cools the battery but cannot stop internal chemical reactions. |
| Foam Fire Suppressants | Designed for hydrocarbon fires, but ineffective for battery electrolyte combustion. |
| Oxygen-Starving Systems | Battery fires generate their own oxygen, making these methods useless. |
Effective Fire Suppression Techniques
To effectively combat lithium-ion battery fires, specialized suppression techniques are required.
1. Water-Based Fire Suppression (Limitations)
🚨 Water is not a perfect solution, but it can help slow the spread of battery fires:
- Cools surrounding areas, preventing further thermal propagation.
- Requires prolonged exposure (sometimes hours) to prevent re-ignition.
- Not effective for deep-seated battery fires.
💡 Best Practice:
Flood EV battery fires with large volumes of water to lower temperatures until thermal runaway stops.
2. Specialized Suppression Systems (NOVEC, FST Aerosol)
🚀 Advanced suppression agents target lithium-ion battery-specific fire risks:
- NOVEC 1230 – A clean agent that absorbs heat quickly while minimizing damage to electronics.
- FST Aerosol Suppression – Releases a fine particulate mist that interrupts combustion chemistry.
💡 Best Practice:
Use in battery storage rooms, data centers, and EV charging stations where traditional extinguishers are ineffective.
3. Gas Extraction Systems
🛑 Removing hazardous gases is crucial for preventing secondary explosions and protecting first responders.
- High-capacity ventilation systems help clear toxic fumes.
- Sealed storage containers with gas extraction prevent pressure buildup.
💡 Best Practice:
Install in industrial battery storage facilities to mitigate toxic gas exposure during failures.
Preventive Safety Measures in Energy Storage and EVs
💡 Preventing fires is always better than fighting them. To reduce the risk of lithium-ion battery fires:
✅ Thermal Monitoring Systems – Early detection of overheating cells.
✅ Battery Management Systems (BMS) – Prevent overcharging and deep discharging.
✅ Fire-Resistant Battery Enclosures – Contain and isolate failed battery cells.
✅ Automatic Gas Ventilation – Prevents toxic buildup in confined spaces.
✅ Emergency Response Training – Educates personnel on proper firefighting techniques for battery fires.
Conclusion
Thermal runaway is a dangerous chain reaction in lithium-ion batteries, leading to fires, explosions, and toxic gas release. It occurs due to overheating, overcharging, short circuits, or mechanical damage.
Preventing thermal runaway requires better battery design, smart management systems, proper handling, and advanced fire suppression methods. Early detection and proactive safety measures can significantly reduce risks in EVs, energy storage, and consumer electronics.
Future improvements include safer cathode materials, advanced insulation, AI-driven monitoring, and stricter safety regulations. As battery technology evolves, enhanced safety measures will be crucial for widespread adoption.
Looking for reliable Battery Testing Equipment? Tertron provides advanced solutions for battery formation, aging, and testing. Improve efficiency and ensure quality with our equipment. Contact us today to discuss your needs and find the right solution.
