Lithium-ion batteries power everything from smartphones to electric vehicles. But did you know not all lithium-ion batteries are the same?
Different types of lithium-ion batteries offer unique benefits, from longer lifespan to higher energy density. Choosing the right one depends on your needs.
In this post, we’ll explore the six main types of lithium-ion batteries. You’ll learn their characteristics, advantages, disadvantages, and best applications.
Fundamentals of Lithium Batteries
What is a Lithium Battery?
A lithium battery is a rechargeable energy storage device that powers various electronic devices, from smartphones to electric vehicles. Unlike traditional lead-acid or nickel-based batteries, lithium-ion batteries are known for their high energy density, long lifespan, and lightweight design.
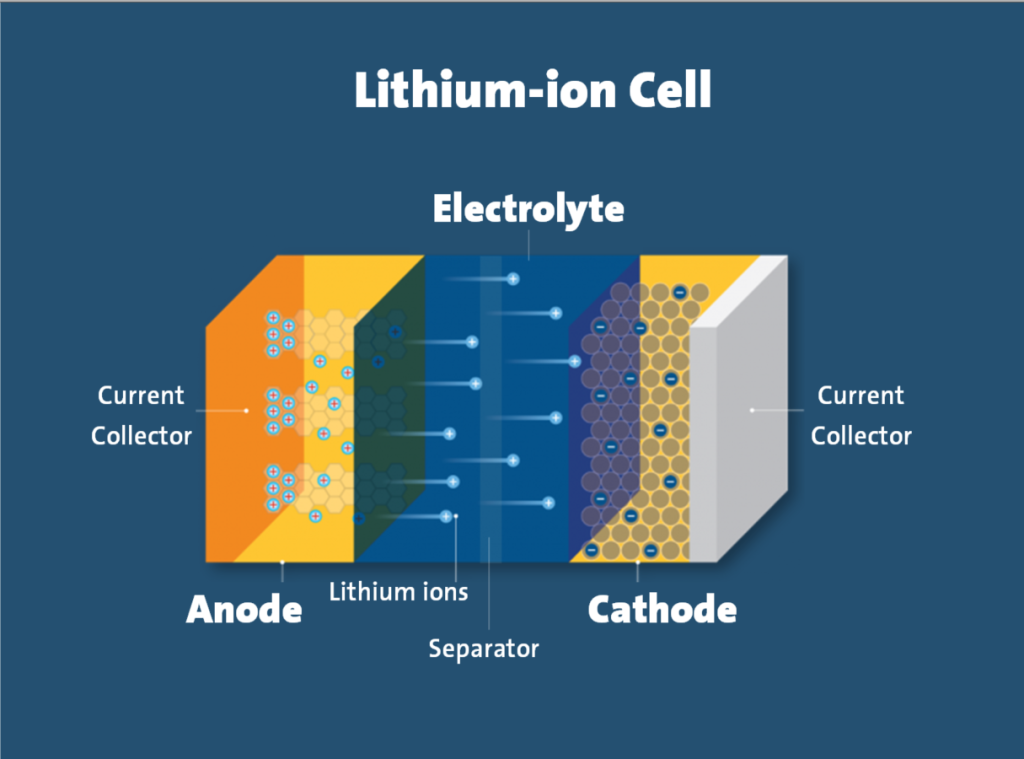
A lithium-ion battery consists of four key components:
- Cathode (positive electrode): Contains lithium compounds that vary by battery type
- Anode (negative electrode): Typically graphite carbon, though some use lithium titanate
- Separator: An insulating layer that allows lithium ions to pass while blocking electrons
- Electrolyte: The medium through which lithium ions travel between electrodes
The working principle of lithium batteries relies on the movement of lithium ions between the cathode and anode. During charging, lithium ions migrate from the positive electrode (cathode) to the negative electrode (anode) through the separator. When discharging, this process reverses, with ions flowing back to the cathode.
This ion movement creates an electrical potential difference, known as voltage. While electrons are blocked by the separator, they’re forced to travel through the external circuit (your device), thus generating the electrical current that powers your electronics.
Classification of Lithium Batteries
Lithium batteries can be classified in two primary ways:
Based on Electrode Materials (Chemistry)
| Type | Cathode Material | Nominal Voltage | Key Advantage | Common Application |
|---|---|---|---|---|
| LFP | Lithium Iron Phosphate | 3.2V | Safety & longevity | Renewable energy storage |
| LCO | Lithium Cobalt Oxide | 3.6V | High energy density | Smartphones, laptops |
| LMO | Lithium Manganese Oxide | 3.7V | High current output | Power tools |
| NMC | Lithium Nickel Manganese Cobalt | 3.6V | Balanced performance | Electric vehicles |
| NCA | Lithium Nickel Cobalt Aluminum | 3.6V | High specific energy | Tesla vehicles |
| LTO | Lithium Titanate | 2.4V | Ultra-fast charging | UPS systems |
Classification by Cell Structure (Form Factor)
Lithium batteries come in different shapes and sizes to fit specific applications:
- Cylindrical Cells – (e.g., 18650, 21700) Used in laptops, EVs, power tools.
- Prismatic Cells – Flat, rectangular design found in smartphones and energy storage systems.
- Pouch Cells – Flexible and lightweight, common in drones, wearables, and automotive applications.
The choice between different lithium battery types depends on the specific requirements of power, energy density, safety, lifespan, and cost considerations.
Lithium Cobalt Oxide (LiCoO2) – LCO
Lithium Cobalt Oxide (LiCoO₂), commonly abbreviated as LCO, is one of the most widely used lithium-ion battery chemistries. It is known for its high energy density, making it ideal for portable consumer electronics.
Composition and Structure
LCO batteries feature a layered structure with:
- Cathode: Lithium cobalt oxide (LiCoO2), containing approximately 60% cobalt
- Anode: Graphite carbon
- Operating Voltage: 3.6V nominal, with a typical range of 3.0-4.2V per cell
Key Characteristics
| Parameter | Value | Notes |
|---|---|---|
| Specific Energy | 150-200 Wh/kg | Specialty cells can reach 240 Wh/kg |
| Cycle Life | 500-1000 cycles | Depends on discharge depth and temperature |
| Thermal Runaway | 150°C (302°F) | Full charge increases thermal runaway risk |
| Charge Rate | 0.7-1C | Higher rates significantly reduce battery life |
| Discharge Rate | 1C maximum | Higher discharge currents shorten cycle life |
Advantages and Disadvantages
Advantages:
- Excellent electrochemical performance
- High energy density in a compact size
- Stable product performance with good consistency
Disadvantages:
- Relatively short lifespan compared to newer chemistries
- Poor thermal stability raises safety concerns
- High manufacturing costs due to expensive cobalt content
- Limited load capability (low specific power)
Primary Applications
LCO batteries have become the standard power source for consumer electronics, including:
- Mobile phones and smartphones
- Tablets and laptops
- Digital cameras and portable gaming devices
- Wearable technology
Despite newer lithium-ion chemistries emerging, LCO batteries remain prevalent in portable electronics where their high energy density outweighs concerns about their shorter lifespan.
Lithium Manganese Oxide (LiMn2O4) – LMO
Lithium Manganese Oxide (LMO) batteries are known for their high power output, improved safety, and cost-effectiveness. They utilize a manganese-based spinel structure, which enhances thermal stability and makes them safer than cobalt-based lithium-ion batteries.
Composition and Structure
- Cathode: Lithium manganese oxide (LiMn2O4) in a three-dimensional spinel structure
- Anode: Graphite carbon
- Structure Benefit: The 3D framework improves ion flow, reducing internal resistance
Key Specifications
| Parameter | Value | Comparison |
|---|---|---|
| Nominal Voltage | 3.7V | Higher than LFP (3.2V) |
| Operating Range | 3.0-4.2V | Standard for most Li-ion |
| Specific Energy | 100-150 Wh/kg | Lower than LCO/NMC |
| Charge Rate | 0.7-1C (up to 3C max) | Good fast-charging capability |
| Discharge Rate | 1C (up to 10C possible) | Excellent for high-power applications |
| Cycle Life | 300-700 cycles | Shorter than most Li-ion types |
| Thermal Runaway | 250°C (482°F) | Much better than LCO (150°C) |
Advantages
- Enhanced Safety: Superior thermal stability compared to cobalt-based batteries
- High Power Delivery: Low internal resistance enables fast charging and high-current discharging
- Design Flexibility: Can be optimized for either longevity, power output, or energy capacity
- Cost-Effective: Uses less expensive manganese instead of costly cobalt
Disadvantages
- Limited Lifespan: Significantly fewer charge cycles than other lithium chemistries
- Moderate Energy Density: Stores about one-third less energy than cobalt-based cells
Primary Applications
LMO batteries excel in applications requiring high-current bursts, including:
- Power tools and cordless equipment
- Medical devices and diagnostic equipment
- Electric vehicle power systems (often blended with NMC)
Many manufacturers now combine LMO with NMC to create hybrid batteries that leverage the high-current capabilities of manganese while gaining the energy density of nickel-manganese-cobalt systems.
Lithium Nickel Manganese Cobalt Oxide (LiNiMnCoO2) – NMC
Lithium Nickel Manganese Cobalt Oxide (NMC) batteries are among the most versatile and widely used lithium-ion chemistries. They offer a balance between high energy density, power output, and lifespan, making them a preferred choice for electric mobility and industrial applications.
Composition and Material Structure
NMC batteries utilize a unique cathode combination:
- Cathode: Lithium nickel manganese cobalt oxide
- Anode: Graphite carbon (some premium versions add silicon for capacity boost)
- Key Insight: The three metals complement each other—nickel provides high energy density but poor stability, manganese offers stability but lower energy, and cobalt combines these benefits.
Performance Specifications
| Parameter | Value | Notes |
|---|---|---|
| Nominal Voltage | 3.6-3.7V | Typical operating range: 3.0-4.2V |
| Specific Energy | 150-220 Wh/kg | Higher than LFP and LMO |
| Cycle Life | 1000-2000 cycles | Excellent longevity |
| Thermal Runaway | 210°C (410°F) | Better safety than LCO |
| Charge Rate | 0.7-1C | Higher rates decrease lifespan |
| Discharge Rate | 1-2C | Good power delivery |
Advantages
- Customizable Performance: Manufacturers can adjust metal ratios to optimize for specific needs:
- NMC111 (⅓ each): Balanced performance
- NMC532/NMC622: Reduced cobalt, higher energy
- NMC811: High nickel content for maximum energy
- Hybrid Capabilities: Delivers both high energy and reasonable power density
- Extended Lifespan: Significantly longer cycle life than LCO or LMO chemistries
Disadvantages
- Cobalt Dependency: Still requires some cobalt, which is expensive and has supply chain concerns
- Nickel Trade-offs: Higher nickel content increases energy but reduces thermal stability
- Cost Considerations: More expensive than LFP batteries
Primary Applications
NMC has become the dominant chemistry for several mobility applications:
- Electric vehicles (often combined with LMO for power bursts)
- E-bikes and electric scooters
- High-performance power tools
- Medical equipment requiring reliable power
As cobalt supplies tighten, manufacturers continue to develop NMC variants with higher nickel content while maintaining acceptable safety margins.
Lithium Iron Phosphate (LiFePO4) – LFP
Lithium Iron Phosphate (LFP) batteries are known for their exceptional safety, long lifespan, and cost-effectiveness. Unlike cobalt-based lithium-ion chemistries, LFP offers thermal stability and durability, making it a preferred choice for electric vehicles and energy storage systems.
Composition and Structure
- Cathode: Lithium iron phosphate (LiFePO4)
- Anode: Graphite carbon electrode
- Operating Voltage: 3.2V nominal (compared to 3.6-3.7V for cobalt-based cells)
Key Performance Metrics
| Parameter | Value | Comparison |
|---|---|---|
| Nominal Voltage | 3.2-3.3V | Lower than other Li-ion types |
| Specific Energy | 90-120 Wh/kg | ~40% less than NMC/NCA |
| Cycle Life | 2,000-5,000+ | 2-5× longer than other lithiums |
| Thermal Runaway | 270°C (518°F) | Extremely stable, even when fully charged |
| Charge Rate | 1C standard | Some cells support faster charging |
| Discharge Rate | 1C continuous, up to 25C pulse | Excellent high-current performance |
| Depth of Discharge | Up to 100% | Can be fully discharged without damage |
Advantages
- Superior Safety Profile
- Inherently stable phosphate chemistry
- Virtually immune to thermal runaway
- No oxygen release during failure
- Exceptional Longevity
- 2,000+ cycle rating at 80% depth of discharge
- Some premium cells exceed 5,000 cycles
- Minimal capacity loss over time
- Practical Benefits
- Tolerant of full discharge conditions
- Excellent performance in high-current applications
- Lower manufacturing costs with abundant materials
Disadvantages
- Lower energy density requires larger battery size for equivalent capacity
- Reduced performance in cold temperatures
- Higher initial cost than lead-acid (though better lifetime value)
Primary Applications
LFP batteries excel in applications where safety, longevity, and cost-effectiveness outweigh size constraints:
- Electric vehicles requiring long service life
- Renewable energy storage systems
- 12V lead-acid battery replacements (four 3.2V cells in series)
- Marine and RV deep-cycle applications
- Uninterruptible power supplies (UPS)
LFP batteries are a reliable, cost-effective, and highly durable choice, particularly for safety-critical applications requiring long-term performance.
Lithium Nickel Cobalt Aluminum Oxide (LiNiCoAlO2) – NCA
Lithium Nickel Cobalt Aluminum Oxide (NCA) batteries are known for their exceptionally high energy density and power output. They are widely used in electric vehicles (EVs) and grid energy storage due to their ability to store large amounts of energy in a compact size.

Composition and Structure
- Cathode Material: Lithium nickel cobalt aluminum oxide (typically contains around 9% cobalt)
- Anode Material: Graphite carbon
- Key Design Philosophy: Aluminum addition to nickel-cobalt oxide provides greater structural stability
Performance Characteristics
| Parameter | Value | Context |
|---|---|---|
| Nominal Voltage | 3.6V | Standard operating range: 3.0-4.2V |
| Specific Energy | 200-260 Wh/kg | Among highest in Li-ion family |
| Projected Potential | Up to 300 Wh/kg | With future refinements |
| Charge Rate | 0.7C typical | Fast charging possible with certain cells |
| Discharge Rate | 1C standard | Higher rates reduce lifespan |
| Cycle Life | ~500 cycles | Significantly less than LFP or NMC |
| Thermal Runaway | 150°C (302°F) | Lower safety margin than other chemistries |
Advantages
- Superior Energy Density: NCA delivers exceptional energy storage capacity by weight
- High Voltage Platform: Maintains stable voltage throughout discharge cycle
- Power Delivery: Good specific power capabilities for acceleration needs
- Extended Service Life: Despite moderate cycle count, maintains good capacity retention
Disadvantages
- Safety Concerns: Among the least thermally stable lithium chemistries
- Temperature Sensitivity: Performance deteriorates at high operating temperatures
- Cost Factors: Expensive due to nickel and cobalt content
- Supply Chain Issues: Reliance on constrained cobalt resources
Primary Applications
- Electric Vehicles: Most notably used by Tesla in their EV battery packs
- Medical Devices: Used in applications requiring high energy in compact form
- Industrial Equipment: Where performance outweighs cost considerations
- Premium Portable Electronics: Where maximum runtime justifies cost
NCA continues to be refined, with ongoing research focused on reducing cobalt content while maintaining performance.
Lithium Titanate (Li4Ti5O12) – LTO
Lithium Titanate (LTO) batteries are known for their exceptional cycle life, rapid charging capabilities, and high safety. Unlike traditional lithium-ion batteries, which use graphite anodes, LTO batteries use lithium titanate, enabling ultra-fast charging and wide temperature operation.

Novel Electrode Configuration
Unlike other lithium-ion batteries that modify the cathode materials, LTO batteries feature:
- Anode: Lithium titanate oxide (Li4Ti5O12) instead of graphite
- Cathode: Typically lithium manganese oxide (LMO) or nickel-manganese-cobalt (NMC)
- Key Difference: The “zero-strain” property of the titanate anode prevents expansion during charging
Performance Profile
| Parameter | Value | Comparison to Other Li-ion Types |
|---|---|---|
| Nominal Voltage | 2.4V | Much lower than standard 3.6-3.7V |
| Specific Energy | 50-80 Wh/kg | ~50% less than LFP, ~70% less than NMC |
| Cycle Life | 3,000-7,000 cycles | 2-3× longer than LFP, 6-10× longer than LCO |
| Thermal Stability | 175-225°C | Extremely safe across all temperature ranges |
| Charge Rate | Up to 10C | Can charge to 80-90% in minutes |
| Discharge Rate | 10C continuous, 30C pulse | Exceptional power delivery |
| Operating Temp | -50°C to +65°C | Widest range of all lithium chemistries |
Key Advantages
- Ultrafast Charging Capability
- Accepts charge rates up to 10C without degradation
- No SEI (Solid Electrolyte Interphase) formation or lithium plating
- Exceptional Longevity
- Can exceed 20,000 cycles at partial discharge levels
- Minimal capacity loss over time
- Extreme Temperature Performance
- Functions in subzero environments where other lithium batteries fail
- No cooling systems required in hot environments
Primary Disadvantages
- Low Energy Density: Requires significantly larger batteries for equivalent capacity
- High Cost: Titanium processing adds substantial expense
- Weight Penalty: Heavier than most lithium-ion options for the same energy
Applications
LTO batteries excel in specialized applications where their unique properties justify the higher cost:
- Electric buses and rapid-transit vehicles (fast-charging at stations)
- Industrial UPS systems requiring exceptional cycle life
- Military and aerospace equipment needing extreme temperature operation
- Solar street lighting with harsh environmental exposure
- Medical devices requiring absolute reliability
Despite higher costs, LTO’s unmatched durability often delivers better lifetime value in demanding applications.
Comparison of Lithium Ion Battery Types
Lithium-ion batteries come in various chemistries, each with unique strengths and tradeoffs. Some prioritize high energy density, while others focus on safety, lifespan, or cost efficiency. Below is a comparison of the key lithium-ion battery types based on critical performance metrics.
Comparative Analysis of Major Lithium Battery Types
| Chemistry | Specific Energy (Wh/kg) | Cycle Life | Thermal Runaway | Relative Cost | Primary Applications |
|---|---|---|---|---|---|
| LCO (Lithium Cobalt Oxide) | 150-200 | 500-1,000 | 150°C (Poor) | High | Smartphones, laptops, cameras |
| LMO (Lithium Manganese Oxide) | 100-150 | 300-700 | 250°C (Good) | Moderate | Power tools, medical devices |
| NMC (Lithium Nickel Manganese Cobalt) | 150-220 | 1,000-2,000 | 210°C (Good) | Moderate-High | EVs, e-bikes, power tools |
| NCA (Lithium Nickel Cobalt Aluminum) | 200-260 | ~500 | 150°C (Poor) | High | Tesla vehicles, grid storage |
| LFP (Lithium Iron Phosphate) | 90-120 | 2,000+ | 270°C (Excellent) | Moderate | Energy storage, buses, marine |
| LTO (Lithium Titanate) | 50-80 | 3,000-7,000 | 175-225°C (Excellent) | Very High | Fast-charging applications, UPS |
Understanding the Critical Trade-offs
The selection of battery chemistry involves balancing five key factors:
- Energy vs. Safety
- High-energy batteries (NCA, LCO) generally have lower thermal stability
- Safer chemistries (LFP, LTO) typically store less energy per unit weight
- Lifespan vs. Initial Cost
- Longer-lasting batteries (LFP, LTO) have higher upfront costs
- Lower-cycle chemistries (LMO, NCA) are cheaper initially but require more frequent replacement
- Power vs. Energy Density
- High-power batteries (LMO, LTO) sacrifice energy density
- Energy-dense options (NCA, LCO) may have limitations on discharge rates
- Temperature Performance
- LFP and LTO excel in extreme temperatures
- NCA and LCO perform best in moderate climate conditions
- Resource Considerations
- Cobalt-heavy chemistries (LCO) face supply constraints and ethical sourcing challenges
- Iron-based (LFP) and titanium-based (LTO) options use more abundant materials
Understanding these fundamental trade-offs helps engineers and consumers select the optimal battery chemistry for their specific application requirements, balancing performance needs against constraints like budget, space limitations, and operational conditions.
Future Developments in Lithium-Ion Batteries
As demand for safer, longer-lasting, and more sustainable lithium-ion batteries grows, researchers and manufacturers are exploring new technologies. Innovations in materials, design, and recycling aim to enhance battery performance while reducing costs and environmental impact.
Key Advancements in Lithium-Ion Battery Technology
🔋 Solid-State Electrolytes – Replacing liquid electrolytes with solid-state materials can increase energy density and safety by eliminating flammable solvents. These batteries promise faster charging, longer lifespan, and lower thermal runaway risks.
🔬 Silicon Anodes for Higher Capacity – Traditional graphite anodes are being replaced or supplemented with silicon-based anodes, which can store up to 10 times more lithium ions. This improvement can significantly boost battery capacity and extend runtime.
🛠 High-Nickel and Nickel-Free Cathodes – Cobalt is expensive and supply-constrained. To reduce reliance on this material, battery manufacturers are developing high-nickel NMC cathodes (e.g., NMC811) and cobalt-free alternatives like lithium iron phosphate (LFP).
♻ Recycling & Sustainable Material Supply Chains – The push for closed-loop battery production focuses on recovering lithium, nickel, and cobalt from used batteries. Advanced recycling methods reduce environmental impact and create a more sustainable battery industry.
Final Thought
Lithium-ion batteries come in various types, each with different energy density, lifespan, and safety levels. Choosing the right one depends on your needs.
Matching the battery type to the application ensures optimal performance, safety, and cost-effectiveness in EVs, energy storage, and electronics.
These batteries power portable devices, electric vehicles, and renewable energy systems, supporting a cleaner and more efficient future.
Ongoing innovations in materials, recycling, and cost reduction will further improve battery technology and accelerate global adoption.
